A new prescription drug takes time and money to develop – on average a whopping 14 years and $2 billion, according to the National Institutes of Health. That’s not happy news for ailing people waiting for the next breakthrough that will bring cure or comfort.
So imagine the pleasure of a researcher at Chapman University School of Pharmacy (CUSP) who helped discover that an inexpensive class of drugs used to treat HIV for 30 years could be repurposed to treat a major cause of blindness among seniors. Even better is that the tried and approved drugs are some the researcher worked on decades ago.

Keykavous Parang, Pharm.D., Ph.D., professor and associate dean of research, graduate studies and global affairs at CUSP, helped uncover a potential new treatment for dry macular degeneration.
“When I started this collaborative work I never thought, ‘Oh, maybe they can use anti-HIV drugs for anti-inflammatory purposes,” said Keykavous Parang, Pharm.D., Ph.D., professor and associate dean of research, graduate studies and global affairs at CUSP.
But Parang and other researchers report that the anti-inflammatory feature of those anti-HIV compounds – called nucleoside reverse transcriptase inhibitors (NRTIs) – could slow or prevent macular degeneration, a leading cause of vision loss worldwide, and one expected to become a growing problem as baby boomers age. Results of the study, led by Dr. Jayakrishna Ambati, professor and vice chair of the Department of Ophthalmology and Visual Sciences at the University of Kentucky, were published recently in
Science.
For HIV patients, the NRTIs, often called “nukes,” have the effect of blocking the virus’s ability to make copies of itself in healthy cells through DNA replication. In macular degeneration, the NRTIs work differently. Through experimentation the scientists found that NRTIs stop an immune response that triggers a chain of proteins to develop – a domino progression that leads to degeneration of the macula. The macula is near the center of the retina and is the part of the eye that provides sharp, central vision.
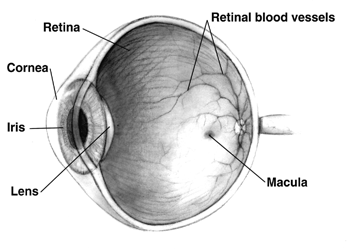
The macula is near the center of the retina and is the part of the eye that provides sharp, central vision.
Parang likened the drug’s mechanism to an interruption in the delivery route of a package sent overseas, with many check-in points along the way to its destination.
“The package has to go through multiple steps,” he says. “But if you block it at one step, that package will not be delivered.”
Further studies need to be done to see how this strategy works in patients, in what form a repurposed version of the drug might be taken and at what stage of the disease it would need to be deployed to be effective, Parang says. But he says toxicity levels, sometimes a concern in HIV patients using NRTIs, will not likely be a problem because the derivatives of the compounds target a different cellular pathway.
Finding new uses for old drugs often happens by serendipity, says Parang, who has a long research career developing HIV, cancer and antibacterial therapies. But in this case it was part of a trend encouraged by the National Institutes of Health
to speed up the creation of new treatments by turning fresh eyes to existing pharmaceuticals. These approved drugs have been tested in humans and come with data on activity, toxicity and formulation, trimming both the time and cost of drug development. Parang is excited by the potential of this movement.
“Everything happens for a reason,” says Parang. “If you have a good eye and you’re in the right place and right time and you have the right people to collaborate with, you can find something useful for treatment of human disorders.”




